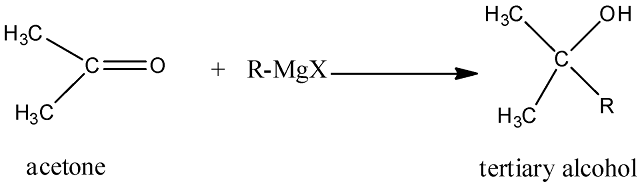Aldehydes, Ketones and Carboxylic Acids Class 12 Notes Chemistry Chapter 12
Introduction
Aldehydes, ketones and carboxylic acids are widespread in plants and animal kingdom. They play an important role in biochemical processes of life. This chapter includes knowledge of carbonyl compounds i.e., the compounds containing >C=O as functional group.
In this chapter, we study aldehydes, ketones, carboxylic acids and their derivatives. In aldehydes, carbonyl group is attached to either two H-atoms or one R- or Ar-group and one H-atom. In ketones carbonyl group is attached to two alkyl or aryl groups.
Nomenclature and Structure of Carbonyl Group
There are two systems of nomenclature of aldehydes and ketones.
(a) Common names
The common names of most aldehydes are derived from the common names of the corresponding carboxylic acids by replacing the ending –ic of acid with aldehyde. The location of the substituent in the carbon chain is indicated by Greek letters α, β, γ, δ, etc. The α-carbon being the one directly linked to the aldehyde group, β-carbon the next, and so on. For example

The common names of ketones are derived by naming two alkyl or aryl groups bonded to the carbonyl group. The locations of substituents are indicated by Greek letters, α α′, β β′ and so on beginning with the carbon atoms next to the carbonyl group, indicated as αα′.
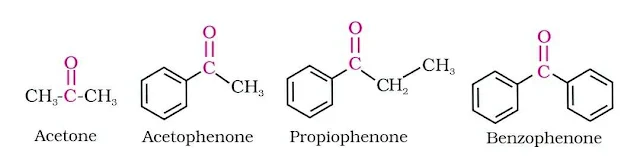
Read also: Amines Chemistry Class 12 Notes Chapter 13
(b) IUPAC names
The IUPAC names of open chain aliphatic aldehydes and ketones are derived from the names of the corresponding alkanes by replacing the ending –e with –al and –one respectively.

Structure of Carbonyl Group
Carbon atom of carbonyl group is sp2 hybridised having triangular planar geometry. Carbon atom forms three sigma bonds and one π(pi) bond, out of which 3σ bonds are located in the same plane whereas fourth π bond, which is formed by lateral or sideways overlapping, is situated above and below the plane. Bond angles are approximately 120°.
Preparation for Aldehydes and Ketones
1. From alcohols
There are two commons ways of converting alcohols to aldehydes or ketones.
(a) By oxidation: Oxidation of alcohols by common oxidants such as K2Cr2O7/H+, KMnO4/H+,KMnO4/OH-, CrO3/H+ gives aldehydes or ketones.
R-CH2-OH + [O] ⟶ R-CHO + H2O
R-CH2(OH)-R + [O] ⟶ R-CO-R
(b) By catalytic dehydrogenation: This method is suitable for volatile alcohols and is of industrial application. In this method alcohol vapours are passed over heavy metal catalysts such as Ag or copper.
R-CH2(OH) ⟶ RCHO + H2
R-CH(OH)-R' ⟶ R-CO-R' + H2
Read also: Atoms Class 12 Physics Notes Chapter 12
2. From acid chlorides
By catalytic hydrogenation of acid chlorides with Pd and BaSO4 poisoned by traces of S or quinoline i.e., Lindlar’s catalyst produces aldehyde. It is also called Rosenmund reduction.
R-CO-Cl + H2 ⟶ R-CHO + HCl
CH3-CO-Cl + H2 ⟶ CH3-CHO + HCl
3. From Hydrocyanic acid
Treatment of hydrocyanic acid with Grignard reagent followed by hydrolysis form aldehyde.
H-C☰N + CH3MgBr ⟶ CH3-CH=N.MgBr ⟶ CH3-CHO + Br-Mg-NH2
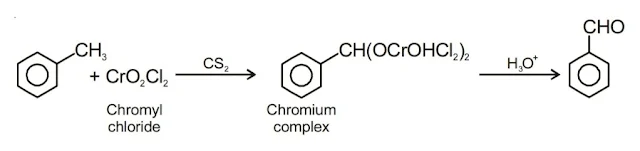
4. Etard reaction
Chromylchloride (CrO2Cl2) Chromyl chloride oxidises methyl group to a chromium complex, which on hydrolysis gives corresponding benzaldehyde. This reaction is called Etard reaction.
Read also: Conceptual Questions for Class 12 Physics Chapter 12 Atoms
5. By Gatterman – Koch reaction
When benzene or its derivative is treated with carbon monoxide and hydrogen chloride in the presence of anhydrous aluminium chloride or cuprous chloride, it gives benzaldehyde or substituted benzaldehyde.
6. From carboxylic acids
By passing vapours of acid over MnO at 573 K.
CH3-CO-OH + HOOC-CH3 ⟶ CO2 + H2O + CH3-CO-CH3
7. From nitriles
Both aliphatic and aromatic ketones can be prepared by treating alkyl or aryl nitrile with suitable Grignard reagent followed by acid hydrolysis.
Physical Properties
1. Physical state: Methanal is a pungent smelling gas, ethanal is volatile liquid with b.p. = 294 K. Rest of the aldehydes and ketones containing upto eleven carbon atoms are colourless liquids while higher members are solids.
2. Solubility: Aldehydes and ketones upto four carbon atoms are miscible with water due to presence of H-bonding between polar carbonyl group and water molecules.
Solubility of these compounds decreases rapidly on increasing length of alkyl chain and higher members are practically insoluble in water. All these compounds are quite soluble in organic solvents such as CH3OH, ether, CHCl3, benzene etc. Ketones, themselves are good solvents.
3. Boiling points: Boiling points of aldehydes and ketones are higher than those of non-polar compounds i.e., hydrocarbons and weakly polar compounds, such as ether, due to dipole-dipole interactions but lower than that of alcohols of comparable molar masses, due to absence of intermolecular H-bondings.
4. Smell: Lower aldehydes have exceptionally unpleasant odour whereas other members generally have pleasant odour. As the size of molecule increases smell becomes less and less pungent and more fragrant.
Chemical Reactions
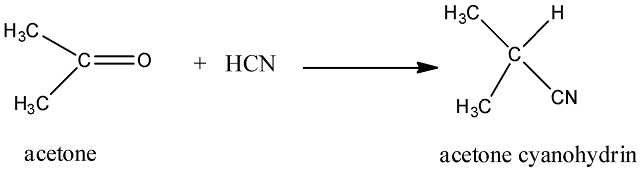
(i) Addition of hydrogen cyanide
Acetaldehyde in reaction with hydrogen cyanide gives acetaldehyde cyanohydrins.
Acetone in reaction with hydrogen cyanide gives acetone cyanohydrins.
(ii) Addition of Grignard reagent
Formaldehyde when reacted with Grignard reagent produces primary alcohol.
Acetaldehyde when reacts with Grignard reagent, it produces secondary alcohol.
Acetone when reacts with Grignard reagent, it produces tertiary alcohol.
(iii) Addition of ammonia derivative
Aldehyde or ketone when reacted with ammonia derivatives forms a group containing −C=N− group.
(iv) Reduction reaction
Aldehydes or ketones on reduction gives respective alcohols in the presence of platinum or palladium and their reactions are given below:
R-CHO ⟶ R-CH2-OH
>C=O ⟶ >CH-OH
The reduction can be done by many catalysts like zinc-amalgam, hydrazine and potassium hydroxide, and hydriodic acid, and red phosphorus.
(v) Oxidation reaction
Aldehydes and ketones on oxidation form respective carboxylic acids.
(vi) Halogenation reaction
Aldehydes and ketones also give halogenations reaction as given below:
(vii) Reaction with alkalis
Aldehydes and ketones in the presence of dilute sodium hydroxide give aldols. These reactions are given below:
Note: The reaction of aldehydes and ketones with alkalies is known as an aldol condensation reaction. Aldehydes and ketones show so many chemical reactions because they are very reactive.
Uses of Aldehydes and Ketones
Aldehydes and ketones are used in chemical industries as solvents, starting material and reagents for synthesising the other products, e.g.,
- 40% aq. HCHO known as formalin and is used in preserving biological specimens.
- Formaldehyde is also used as disinfectant.
- HCHO is used in manufacturing synthetic polymers like Bakelite.
- Acetaldehyde is used for silvering of mirrors.
- Benzaldehyde as a flavouring agent in perfume industry.
- Acetone is used as a solvent in industries and laboratories.
- Acetone is a common constituent of nail polishes. Many aldehydes and ketones such as butyraldehyde, vanilin, acetophenone, camphor etc. are known for their odours and flavours.
Nomenclature of Carboxyl Group
Common names of carboxylic acids are based on their source of origin e.g., Formic acid (HCOOH) was first obtained from red ants (Latin Formica means ants) similarly acetic acid (CH3COOH) is so named because it was obtained from vinegar (Latin Acetum means vinegar) butyric acid (CH3CH2CH2COOH) from rancid butter (Latin Butyrum means butter) etc. In common system position of substituents is indicated by Greek letters α, β, γ, δ etc.
As per IUPAC system, the name of acid is derived by replacing suffix ‘e’ of alkane by ‘oic acid’. Position of substituents is indicated by following rules:
- Select longest chain containing the carboxylic group as parent chain.
- While numbering the chain, the carbon of carboxylic group is numbered one.
- Position of substituents is indicated by the number.
- When compound contains more than one carboxylic group the suffix ‘e’ of alkane is retained and prefixes di, tri or tetra are used to the term oic acid.
- Position of —COOH group is indicated by arabic numerals before the “oic acid”.
Structure of Carboxyl Group
Methods of Preparation of Carboxylic Acids
1. From primary alcohols
Primary alcohols are easily oxidised to carboxylic acids with oxidants such as KMnO4, in neutral, acidic or alkaline media or by CrO3 and K2Cr2O7 in acidic media.
CH3-CH2-OH ⟶ CH3COOH
2. From oxidation of aldehydes and ketones
Aldehydes can be oxidised to carboxylic acids even with mild oxidants while ketones are oxidised with difficulty and usually mixture of acids is obtained.
CH3CHO ⟶ CH3COOH
CH3COCH3 ⟶ CH3COOH + CO2 + H2O
3. By hydrolysis of nitriles (cyanides) and amides
Nitriles are hydrolysed to amides and then to carboxylic acids in the presence of H+ or OH- as catalyst. If conditions used are mild, then reaction can be stopped even at amide.
R-CN + 2H2O ⟶ RCONH2 ⟶ RCOOH + NH3
4. From Grignard reagent
Reaction of Grignard reagent with dry ice (i.e., solid CO2) in ethereal solution followed by acidic hydrolysis produces carboxylic acid containing one more carbon atom than Grignard reagent.
5. From hydrolysis of acid halides and anhydrides
Acid chlorides and anhydrides are readily hydrolysed with aqueous base to form carboxylate ions which form carboxylic acid on acidification.
RCOCl ⟶ RCOOH + HCl
Physical Properties
1. Physical State: First 3 members are colourless liquids with pungent smell, next 6 members are oily liquids with unpleasant odour. Higher acids are colourless waxy solids. Benzoic acid and its homologues are colourless solids.
2. Solubility: Among aliphatic acids first four members are very soluble in water and solubility decreases gradually with rise of molecular mass. All are soluble in alcohol or ether. Benzoic acid is sparingly soluble in cold water but is soluble in hot water, alcohol, ether.
3. Boiling Point: Due to the presence of H-bond, boiling points of carboxylic acids are higher than corresponding aldehydes, ketones, ethers or even alcohols of comparable molar masses.
4. Melting Point: Melting point of carboxylic acid increases irregularly with increasing molar mass, upto first ten numbers, melting point of carboxylic acids containing even number of carbon atoms is higher than the next lower and higher member containing odd number of carbon atoms.
Chemical Reactions
1. Reaction with Metals
The reaction of carboxylic acids with metals like K, Na, Mg, and Ca results in the formation of salts. A proton will be released from the carboxyl group of the carboxylic acid where the metal substation will occur during the reaction phase. H2 gas is produced as a result of the reaction.
2CH3COOH + 2Na ⟶ 2CH3COONa + H2
2. Reaction with Alkalis
The reaction of carboxylic acids with alkalis results in the formation of salts and water.
CH3COOH + NaOH ⟶ CH3COONa + H2O
3. Reaction with Carbonates and Bicarbonates
Carboxylic acids decompose with carbonates and bicarbonates to produce salts, vapor, and carbon dioxide gas.
2CH3COOH + Na2CO3 ⟶ 2CH3COONa + H2O + CO2
2CH3COOH + NaHCO3 ⟶ CH3COONa + H2O + CO2
4. Formation of Acyl Chlorides
Acid chlorides are formed when carboxylic acids react with thionyl chloride (SOCl2), phosphorus pentachloride (PCl5), or phosphorus trichloride (PCl3).
RCOOH + SOCl2 ⟶ RCOCl + SO2 + HCl
RCOOH + PCl5 ⟶ RCOCl + PCl3 + HCl
3RCOOH + PCl3 ⟶ 3RCOCl + H3PO3
5. Formation of Esters (Esterification)
When carboxylic acids and alcohols are heated in the presence of concentrated sulphuric acid or dry hydrochloric acid, fruity esters are formed.
R-COOH + R'OH ⇌ RCOOR' + H2O
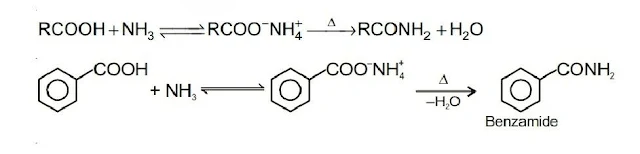
6. Formation of Amide
Ammonium salts are formed by treating carboxylic acids with ammonia. Ammonium salts lose a water molecule when heated, resulting in the formation of amides.
7. Decarboxylation
Decarboxylation reaction occurs when soda lime (NaOH + CaO) is distilled with sodium salts of carboxylic acids, resulting in alkanes.
R-COONa + NaOH ⟶ RH + Na2CO3
CH3-COONa + NaOH ⟶ CH4 + Na2CO3
8. Formation of Anhydrides
Acid anhydrides are formed when two molecules of carboxylic acid are heated with a dehydrating agent such as phosphorus pentoxide.
9. Hell-Volhard Zelinsky Reaction
Carboxylic acids shape -substituted carboxylic acids when they react with a chlorine (Cl2) or bromine (Br2) molecule. In the presence of red phosphorus, the reaction takes place. The H.V.Z. reaction, or Hell-Volhard Zelinsky reaction, is the name given to this reaction.
10. Ring substitution in aromatic acids
–COOH group in aromatic carboxylic acids is a ring deactivating meta directing group. They do not undergo Friedel-Craft reaction, because carboxyl group is deactivating and catalyst AlCl3 (Lewis acid) gets bonded to carbonyl group. Some common electrophilic substitution reactions of benzoic acid are as following:
Uses of Carboxylic Acids
- Methanoic acid is used in rubber, textile, dyeing, leather and electroplating industries.
- Ethanoic acid is used as solvent and as vinegar in food industry. Hexanedioic acid is used in the manufacture of nylon-6, 6.
- Esters of benzoic acid are used in perfumery.
- Sodium benzoate is used as a food preservative.
- Higher fatty acids are used for the manufacture of soaps and detergents.